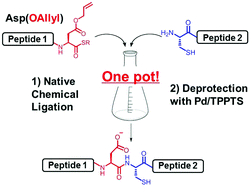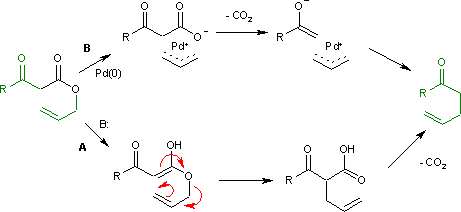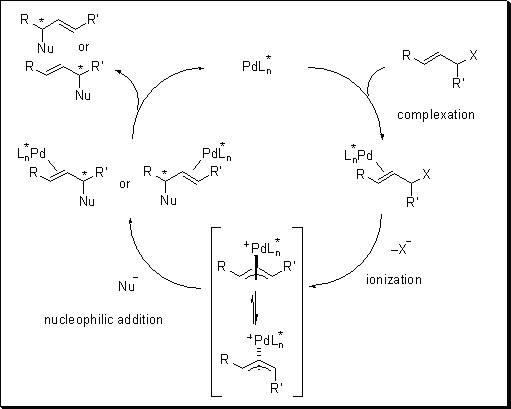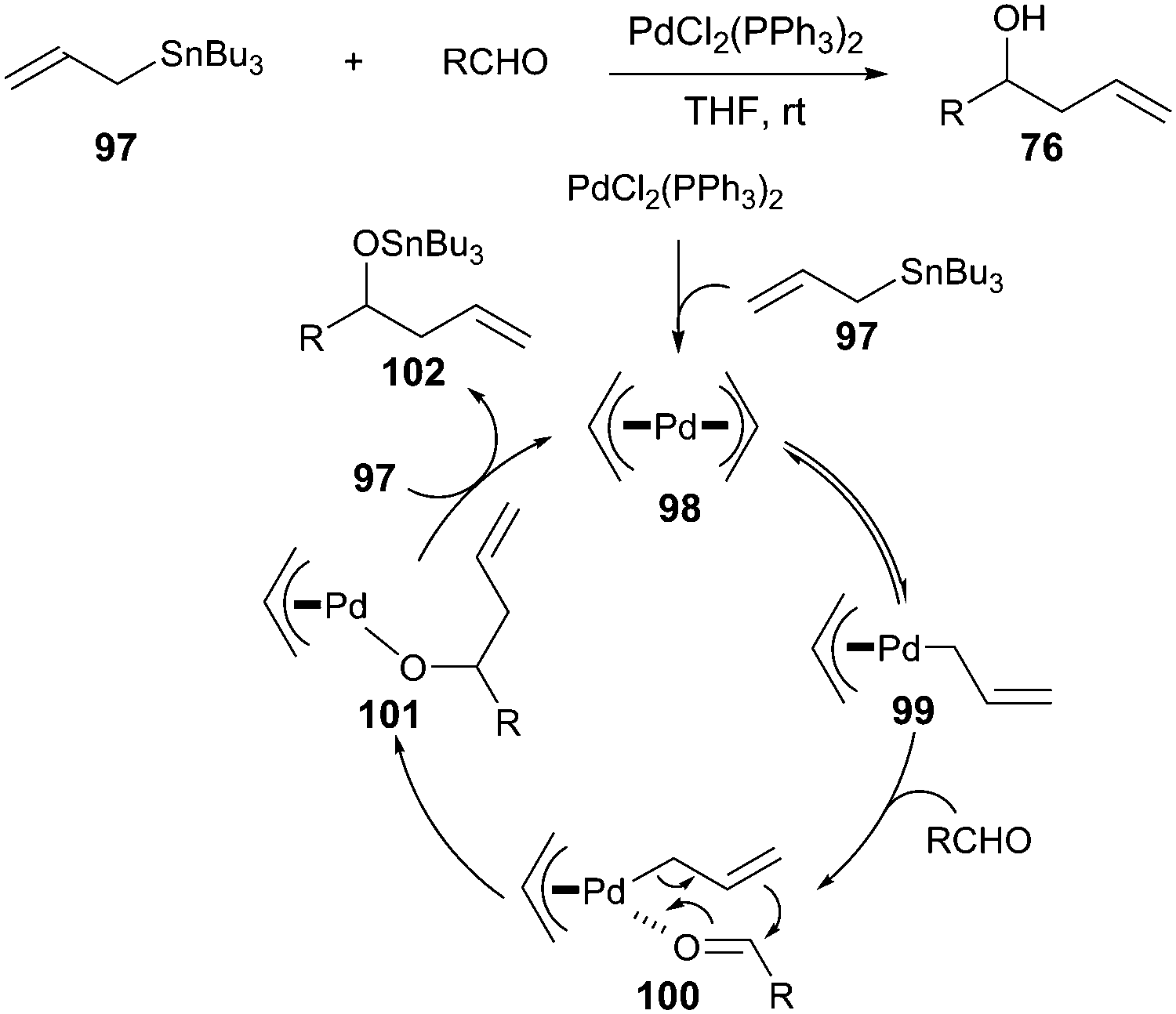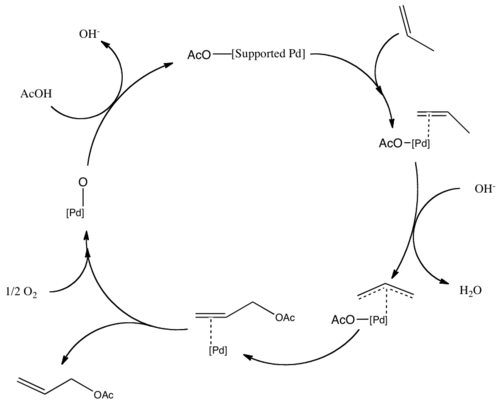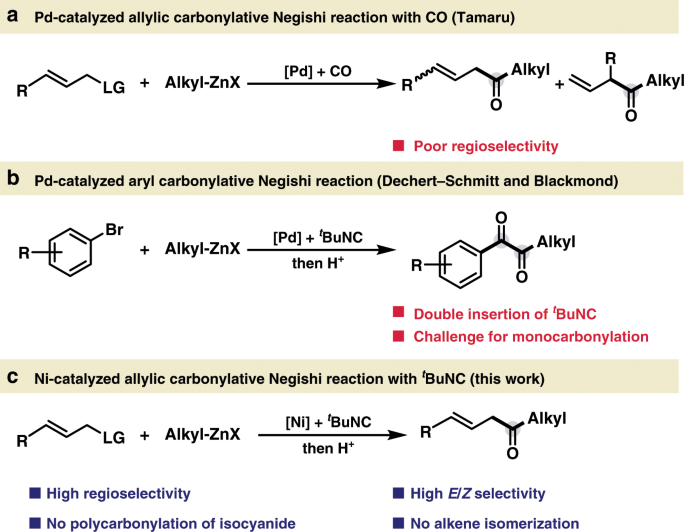
Nickel-catalyzed allylic carbonylative coupling of alkyl zinc reagents with tert -butyl isocyanide | Nature Communications

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane-ZnCl2/Pd(PPh3)4

Irreversible Catalytic Ester Hydrolysis of Allyl Esters to Give Acids and Aldehydes by Homogeneous Ruthenium and Ruthenium/Palladium Dual Catalyst Systems - Nakamura - 2011 - Advanced Synthesis & Catalysis - Wiley Online Library

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane–ZnCl2/Pd(PPh3)4 - ScienceDirect
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions
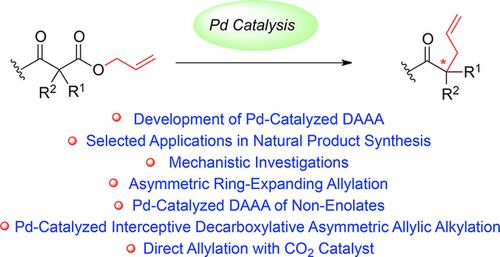
Palladium‐Catalyzed Decarboxylative Asymmetric Allylic Alkylation: Development, Mechanistic Understanding and Recent Advances,Advanced Synthesis & Catalysis - X-MOL
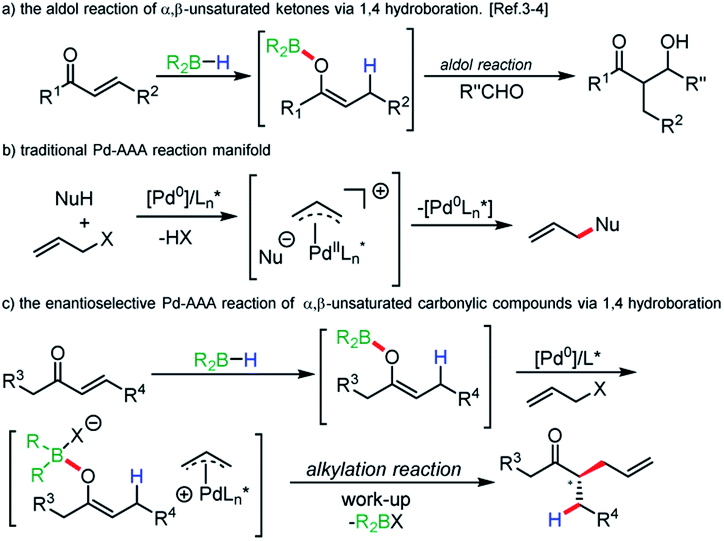
A borane-mediated palladium-catalyzed reductive allylic alkylation of α,β-unsaturated carbonyl compounds - Chemical Science (RSC Publishing) DOI:10.1039/C9SC05970A
Research Letter A Novel, One-Step Palladium and Phenylsilane Activated Amidation from Allyl Ester on Solid Support

Safe Removal of the Allyl Protecting Groups of Allyl Esters using a Recyclable, Low‐Leaching and Ligand‐Free Palladium Nanoparticle Catalyst - Takagi - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library
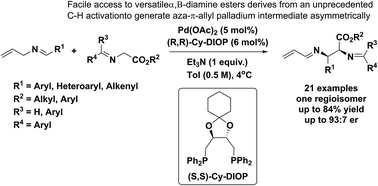
Pd-catalyzed asymmetric allylic alkylations via C–H activation of N-allyl imines with glycinates - Chemical Science (RSC Publishing)