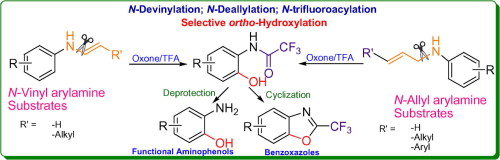
Direct C–N bond cleavage of N-vinyl or N-allyl arylamines: a metal-free strategy for N-devinylation and N-deallylation - Tetrahedron Lett. - X-MOL

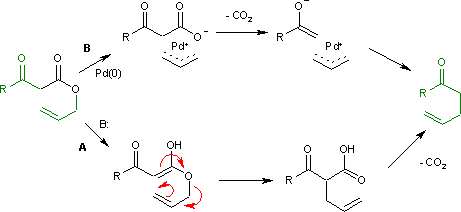
Palladium-Catalyzed Synthesis of Substituted Cycloheptane-1,4-diones by an Asymmetric Ring-Expanding Allylation (AREA)

Noneffervescent Method for Catalysis-Based Palladium Detection with Color or Fluorescence,ACS Sensors - X-MOL

Synthesis of 2-substituted indoles via a palladium-catalyzed domino Heck reaction and dealkylation - ScienceDirect
Palladium-Catalyzed Oxidative N-Dealkylation/Carbonylation of Tertiary Amines with Alkynes to α,β-Alkynylamides

Overcoming the Deallylation Problem: Palladium(II)-Catalyzed Chemo-, Regio-, and Stereoselective Allylic Oxidation of Aryl Allyl Ether, Amine, and Amino Acids.,Organic Letters - X-MOL

N-allyl-N-sulfonyl ynamides as synthetic precursors to amidines and vinylogous amidines. An unexpected N-to-C 1,3-sulfonyl shift in nitrile synthesis. - Abstract - Europe PMC
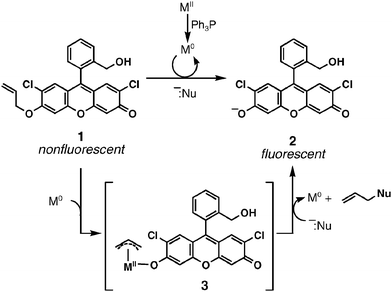
Studies of a fluorogenic probe for palladium and platinum leading to a palladium-specific detection method - Chemical Communications (RSC Publishing)
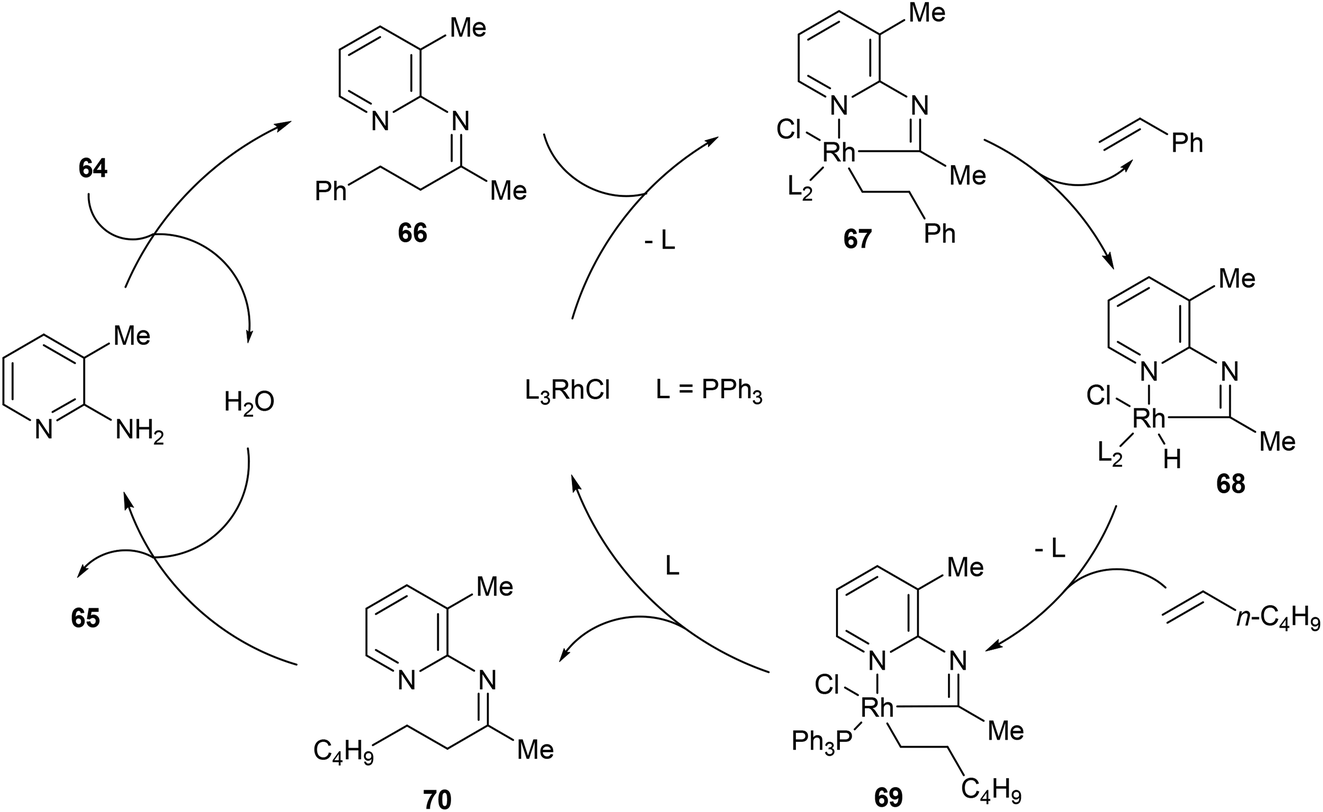
Direct activation of relatively unstrained carbon–carbon bonds in homogeneous systems - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C4QO00053F

Fullerene-bearing porous polymer via ball-milling approach and its palladium composite for catalytic deallylation - ScienceDirect
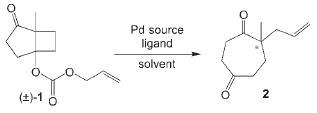
Palladium-Catalyzed Synthesis of Substituted Cycloheptane-1,4-diones by an Asymmetric Ring-Expanding Allylation (AREA)
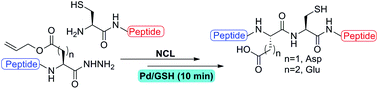
Palladium mediated deallylation in fully aqueous conditions for native chemical ligation at aspartic and glutamic acid sites - Organic & Biomolecular Chemistry (RSC Publishing)
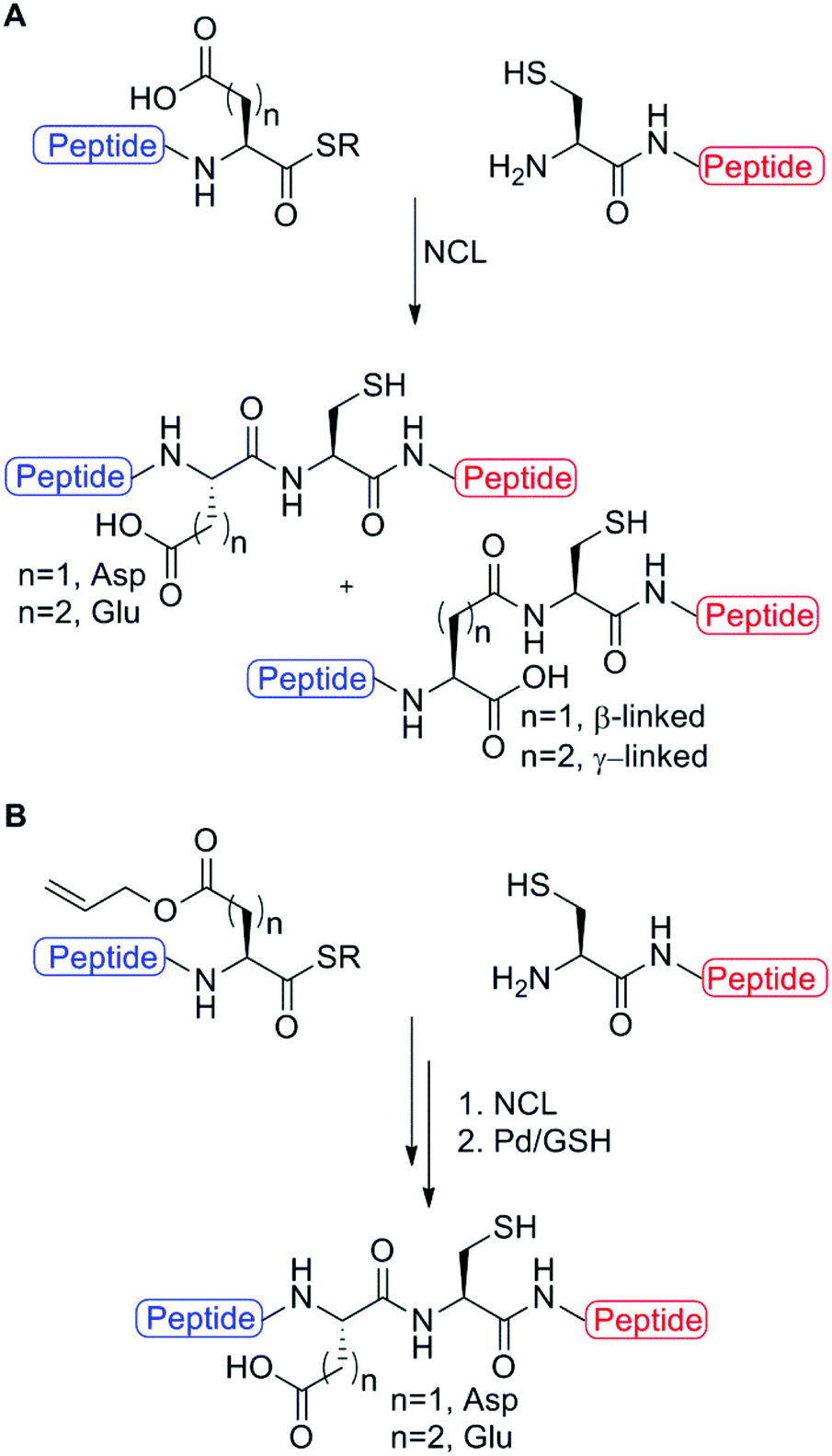
Palladium mediated deallylation in fully aqueous conditions for native chemical ligation at aspartic and glutamic acid sites - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB00890F

Catalysts | Free Full-Text | PdI2-Based Catalysis for Carbonylation Reactions: A Personal Account | HTML