
Rigid tetrazine fluorophore conjugates with fluorogenic properties in the inverse electron demand Diels–Alder reaction - Organic & Biomolecular Chemistry (RSC Publishing)

Inverse-electron-demand Diels–Alder reactions between tetrazines and... | Download Scientific Diagram

Figure 2 from Clicking 1,2,4,5-tetrazine and cyclooctynes with tunable reaction rates. | Semantic Scholar
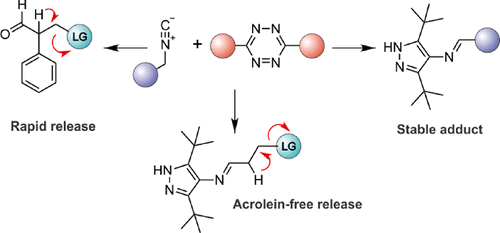
Tuning Isonitrile/Tetrazine Chemistry for Accelerated Deprotection and Formation of Stable Conjugates. - J. Org. Chem. - X-MOL
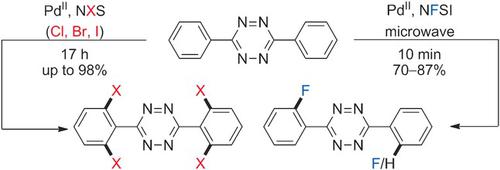
Ortho‐Functionalized Aryltetrazines by Direct Palladium‐Catalyzed C−H Halogenation: Application to Fast Electrophilic Fluorination Reactions,Angewandte Chemie International Edition - X-MOL

Tetrazine‐Triggered Release of Carboxylic‐Acid‐Containing Molecules for Activation of an Anti‐inflammatory Drug - Davies - 2019 - ChemBioChem - Wiley Online Library
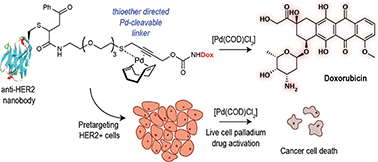
A thioether-directed palladium-cleavable linker for targeted bioorthogonal drug decaging - Chemical Science (RSC Publishing)

Tetrazine‐Triggered Release of Carboxylic‐Acid‐Containing Molecules for Activation of an Anti‐inflammatory Drug - Davies - 2019 - ChemBioChem - Wiley Online Library
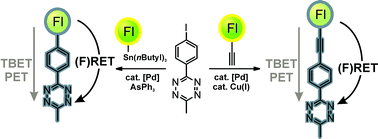
Rigid tetrazine fluorophore conjugates with fluorogenic properties in the inverse electron demand Diels–Alder reaction - Organic & Biomolecular Chemistry (RSC Publishing)

Investigation of activity, stability, and degradation mechanism of surface-supported Pd-PEPPSI complexes for Suzuki-Miyaura coupling - ScienceDirect
![PDF] s-Tetrazines functionalized with phenols: synthesis and physico-chemical properties | Semantic Scholar PDF] s-Tetrazines functionalized with phenols: synthesis and physico-chemical properties | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7dfee7c7d2f52fd26b61fae4ddaa7b589b3539a4/24-Figure32-1.png)
PDF] s-Tetrazines functionalized with phenols: synthesis and physico-chemical properties | Semantic Scholar
![1,2,4,5-Tetrazines and Azolo[1,2,4,5]tetrazines: Synthesis and Reactions with Nucleophiles | SpringerLink 1,2,4,5-Tetrazines and Azolo[1,2,4,5]tetrazines: Synthesis and Reactions with Nucleophiles | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs10593-013-1232-2/MediaObjects/10593_2013_1232_Fig2_HTML.gif)
1,2,4,5-Tetrazines and Azolo[1,2,4,5]tetrazines: Synthesis and Reactions with Nucleophiles | SpringerLink

New heteroaromatic aminations on 5-aryl-1,2,4-triazines and 1,2,4,5- tetrazines by palladium catalysis - ScienceDirect
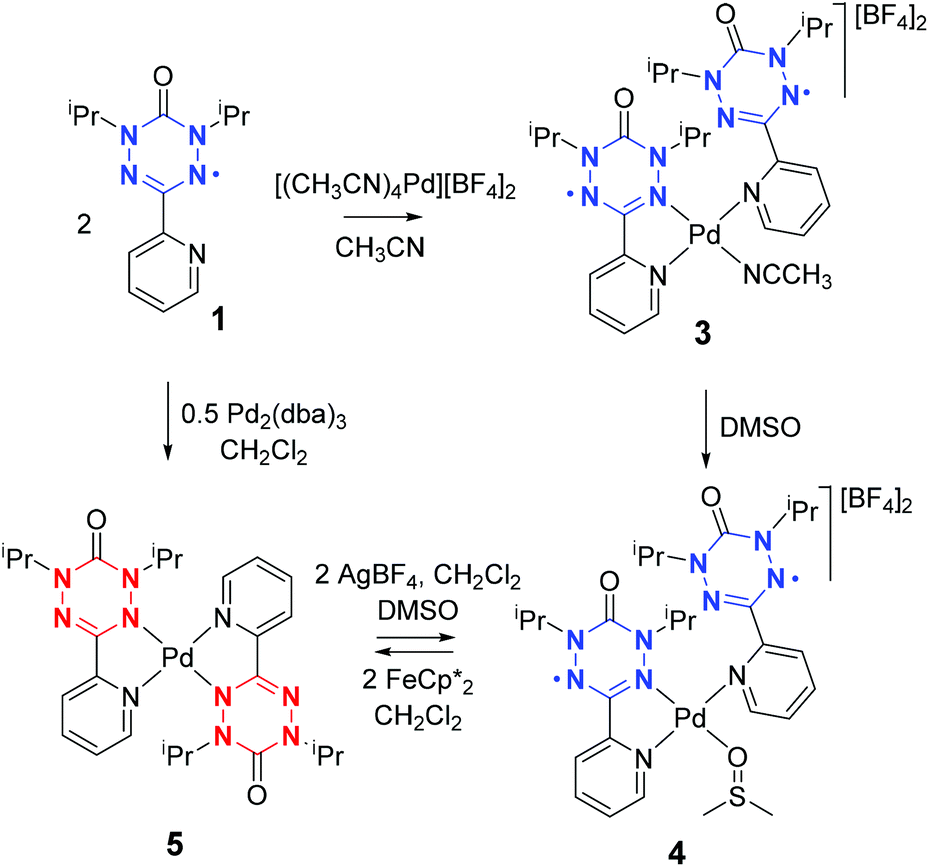
Synthesis and redox reactions of bis(verdazyl)palladium complexes - Dalton Transactions (RSC Publishing)









